|
In the case of period 2, there are two core electrons in the #"1s"# subshell.įor the transition metals, determining valence electrons is tricky because they can use inner electrons as valence electrons. The rest of the electrons are the core electrons. 10":#"1s"^2"2s"^2"2p"^6"#įor all of the preceding elements, the valence electrons are the outermost (highest energy) #"s"# and #"p"# electrons. The Shorthand electron configuration (or Noble gas configuration) as well as Full electron configuration is also mentioned in the table. In 1869 Russian chemist Dmitri Mendeleev. Electron configuration chart of all Elements is mentioned in the table below. Meanwhile, elements in the same period have the same number of occupied electron shells. Elements in the same group have the same number of valence electrons. A neutral atom has the same number of electrons as protons. The periodic table is organized into groups (vertical columns), periods (horizontal rows), and families (groups of elements that are similar). The atomic number is the number of protons in the nuclei of the atoms of an element. (b) Covalent radii of the elements are shown to scale. 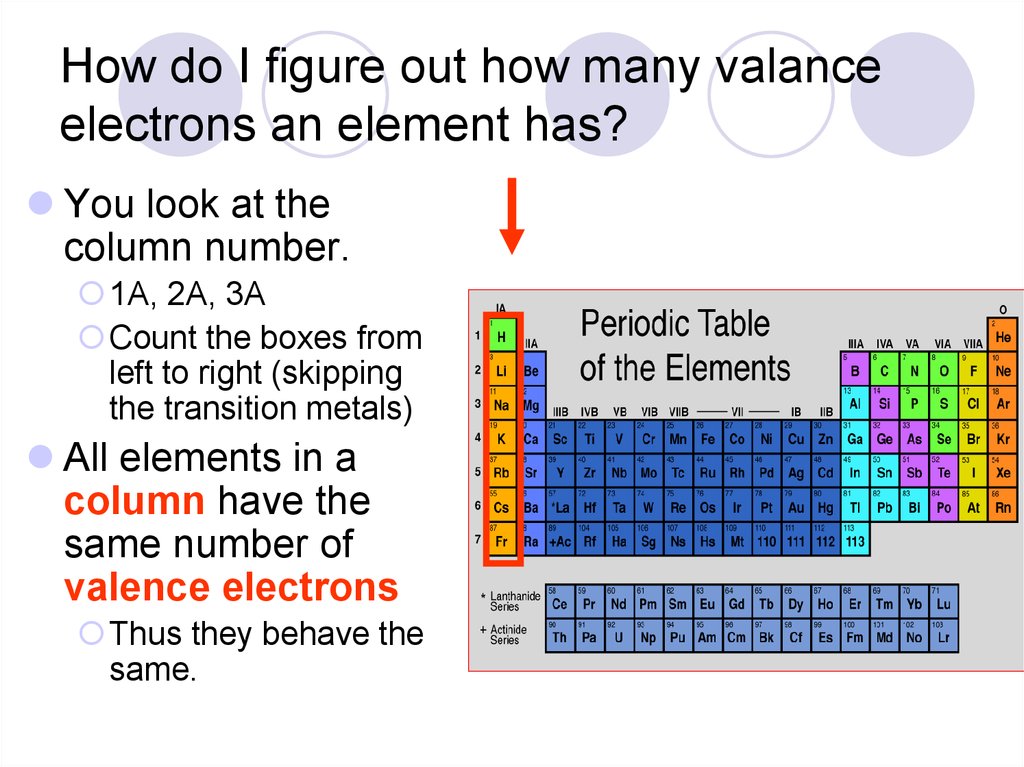
The atomic radius for the halogens increases down the group as n increases. You can also find the core and valence electrons by determining or looking up the electron configurations of the main group elements. 1: (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. 9.9: Periodic Trends - Atomic Size, Ionization. The chemical properties of elements are determined primarily by the number and distribution of valence electrons. Electron configurations can be predicted by the position of an atom on the periodic table. 
Across a period, elements in group 1/IA have one valence electron, elements in group 2/IIA have two valence electrons, elements in group 13/IIIA have three valence electrons, and so on, ending with group 18/VIIIA, which have eight valence electrons, which is the maximum number of valence electrons. The arrangement of electrons in atoms is responsible for the shape of the periodic table. You can determine the number of valence electrons in the atoms of the main group elements by the group number of the element. The core electrons are in the inner shells and do not participate in chemical reactions. The main group elements are the A groups, or groups 1,2,13-18. 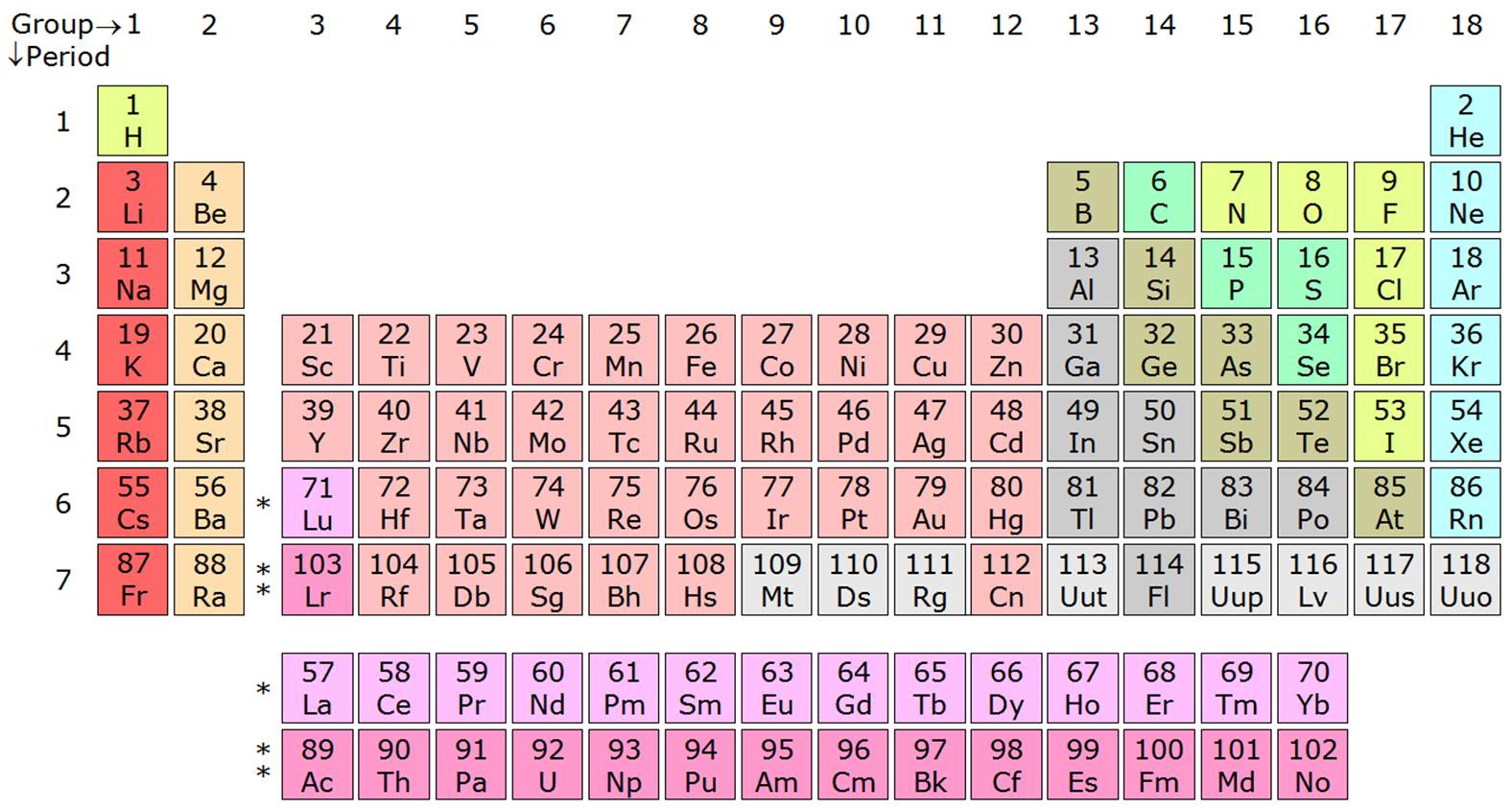
The number of valence electrons in one atom of each element is easily determined based on its position in the periodic table. The valence electrons participate in chemical reactions. Valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation of chemical bonds. In order to determine if an atom/ion is paramagnetic or diamagnetic, we must look at its orbital. Also, notice that as you move down a column, the number of electrons in the outermost principle energy level. aluminum oxide, Al 2 O 3: aluminum has a valence of 3. Valence Electrons and the Periodic Table We saw that Mendeleev arranged elements with similar periodic properties in the same column. 
hydrogen sulfide, H 2 S: sulfur has a valence of 2. Solid nonmetals are also very brittle.For the main group (representative) elements, the valence electrons are the outermost (highest energy) #"s"# and #"p"# electrons, which make up the valence shell. lithium oxide, Li 2 O: lithium has a valence of 1. A nonmetal is typically dull and a poor conductor of electricity and heat. Metals are also malleable (they can be beaten into thin sheets) and ductile (they can be drawn into thin wires). A metal is a substance that is shiny, typically (but not always) silvery in color, and an excellent conductor of electricity and heat.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
